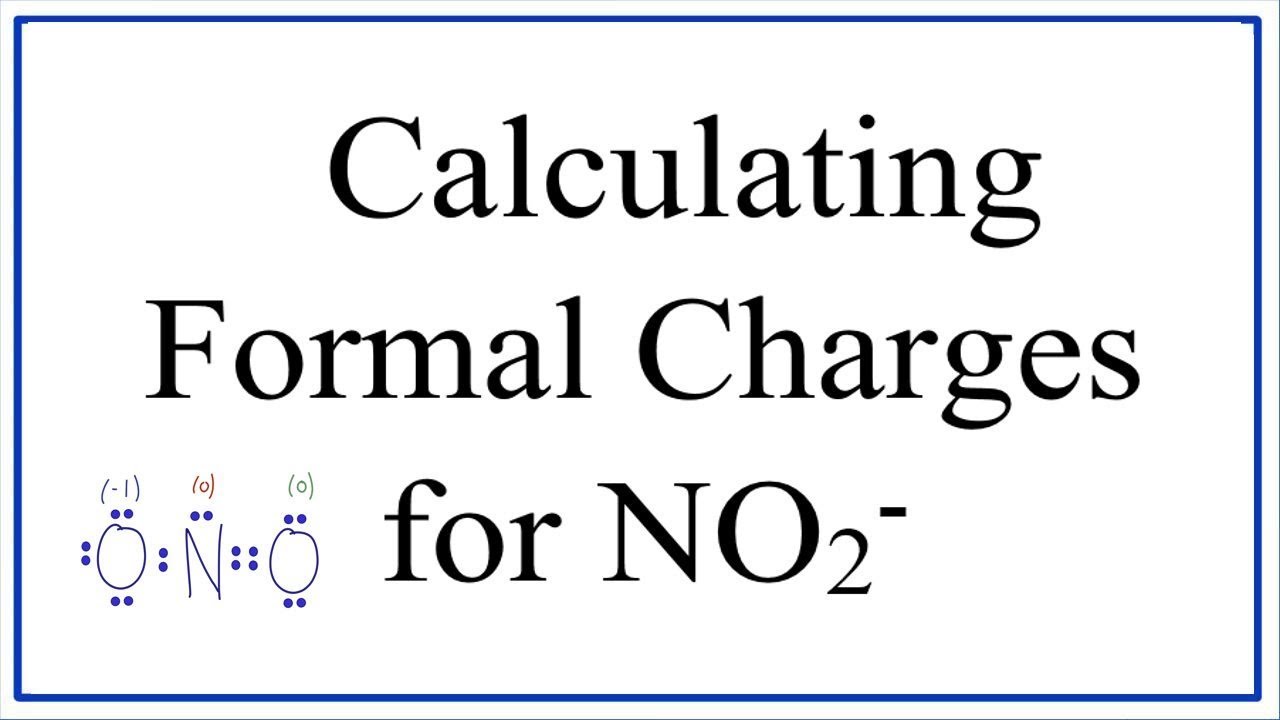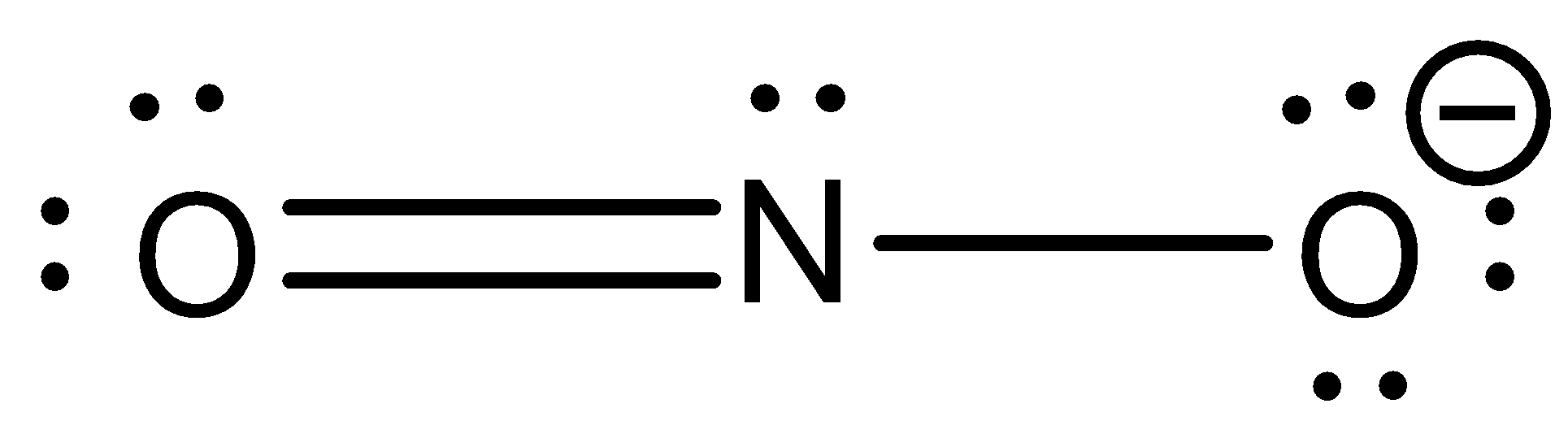NO2 can react with the NO in smog, forming a bond between the N atoms. Draw the structure of the resulting - brainly.com

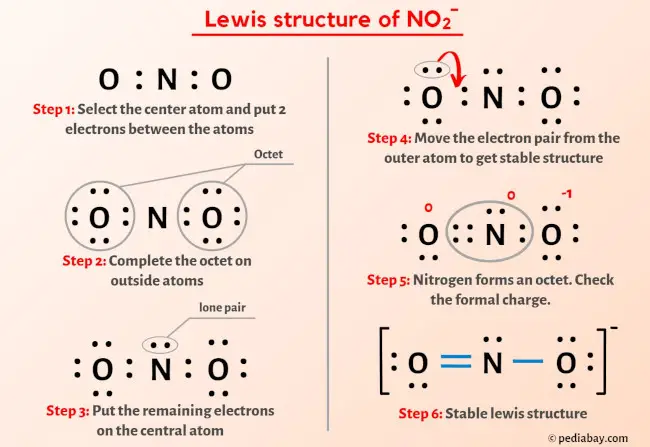
Lewis Electron - Dot Structure of the nitrite ion NO2- | Chemistry classroom, Chemistry education, Chemistry notes

Draw both resonance forms of the nitrite ion, NO_2^- Be sure to include all lone pairs and non-zero formal charges. | Homework.Study.com

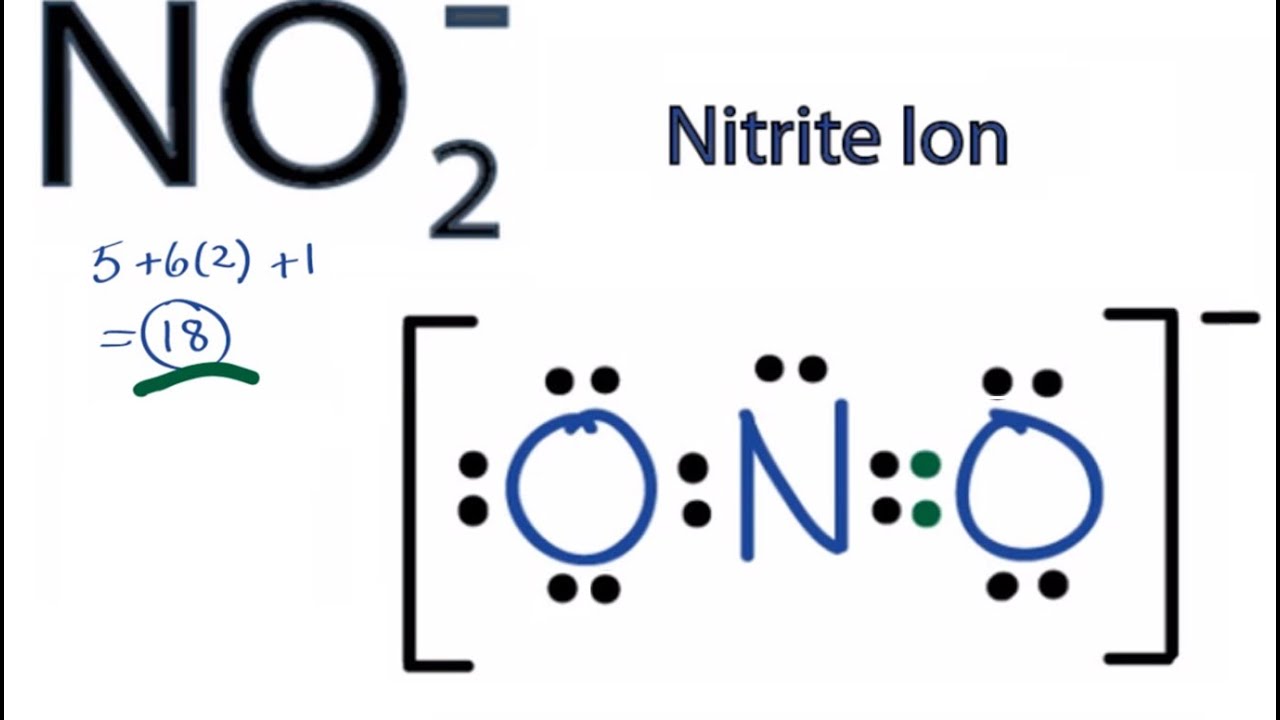
NO2- Lewis Structure (Nitrite Ion) | NO2- Lewis Structure (Nitrite Ion) Hello Everyone! Welcome back to Geometry of Molecules, and for today's video, we will help you with the Lewis... | By

Draw both resonance forms of the nitrite ion, NO_2^- Be sure to include all lone pairs and non-zero formal charges. | Homework.Study.com

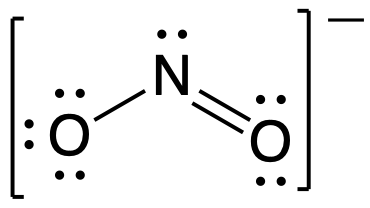
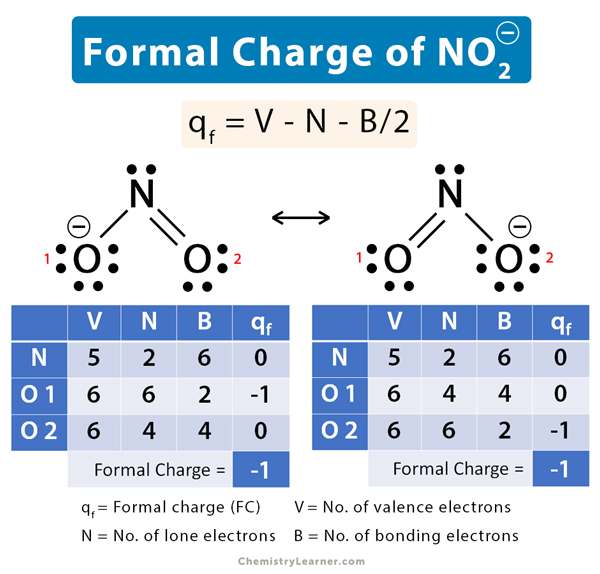
Complete the three Lewis structures for N(NO2)2– by adding missing lone pair electrons and assigning formal - brainly.com

![Solved The dinitramide anion [N(NO2)2 ] was first isolated | Chegg.com Solved The dinitramide anion [N(NO2)2 ] was first isolated | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F163%2F16312270-8746-47ad-92f1-3b9d2989ae86%2FphpWFgMet.png)